Type 1 Diabetes Research 2021: Science, Hope and Clinical Reality
By Natalie SainzMatthew Garza
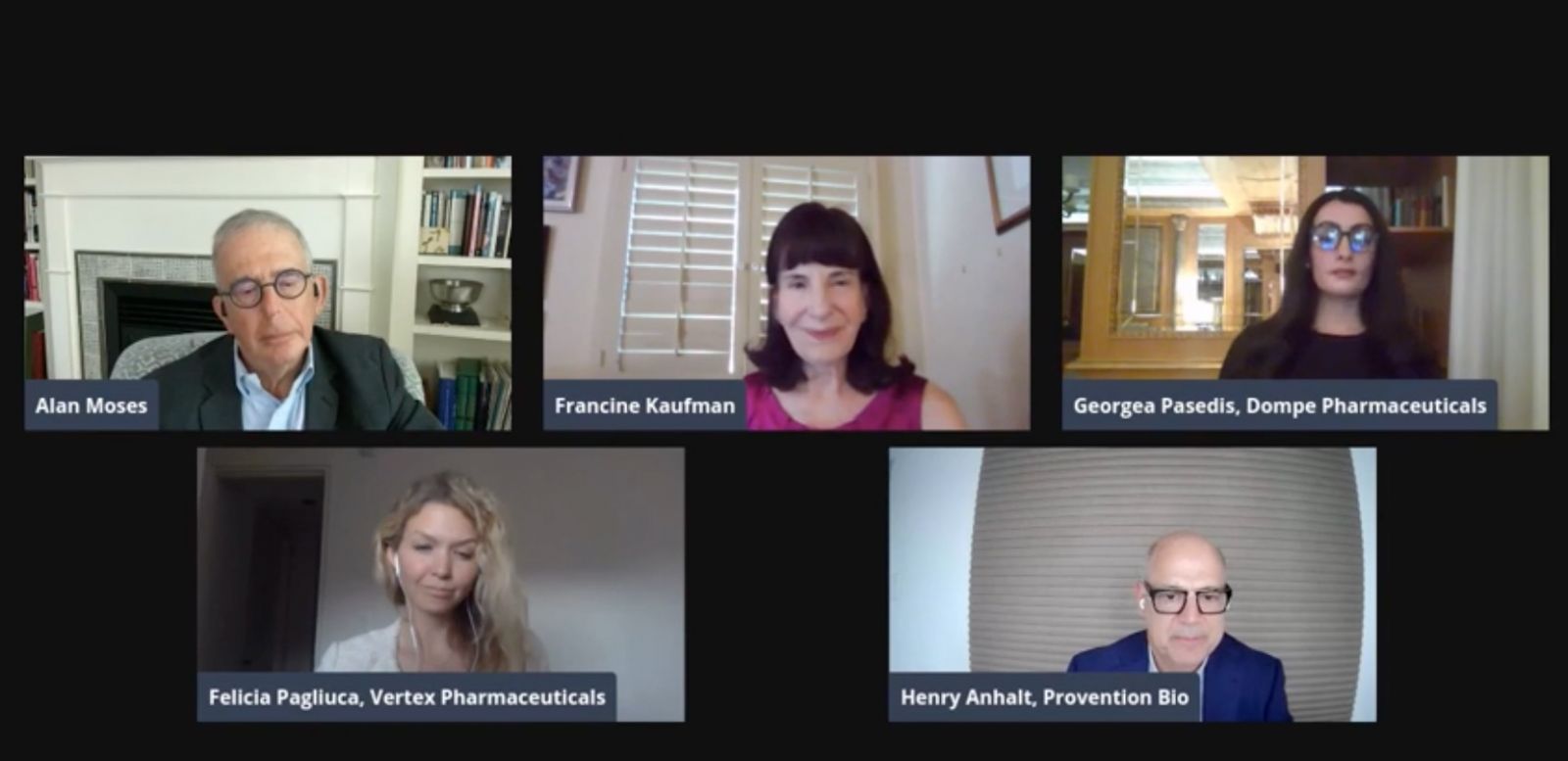 At our second virtual Musings event of the year, we heard five experts discuss type 1 diabetes preventions, delays, and cures – including the importance of screening.
At our second virtual Musings event of the year, we heard five experts discuss type 1 diabetes preventions, delays, and cures – including the importance of screening.
Diabetes treatment has come a long way since the discovery of insulin 100 years ago. In recent years, we have seen major advancements, including those in next-generation basal insulin, immunosuppressive medications to delay type 1 diabetes, automated insulin delivery (AID), and emergency rescue glucagon, all of which have altered the ways people live with diabetes. With each day, researchers are getting closer to the possibility of preventing, delaying, and curing type 1 diabetes, and the new therapies and technology that have been recently developed are helping people with diabetes live longer, healthier lives.
However, it is important to remember that every new potential treatment requires clinical trials to test its safety and effectiveness. These trials take time and require a large, diverse group of participants to get the most representative and accurate results possible. Clinical trials have the potential to transform the lives of many thousands of people at risk for type 1 diabetes and people who already have the disease.
On July 14, diaTribe hosted its second Musings event of the year: “Type 1 Diabetes Research 2021: Science, Hope, and Clinical Reality.” The virtual event drew over 300 participants who engaged in a lively discussion on the future of diabetes clinical trials and treatments – including prevention and cures. You can view the event here.
The discussion featured four experts and was moderated by diaTribe board member Dr. Alan Moses:
-
Dr. Henry Anhalt, Pediatric Endocrinologist & Executive Director of Medical Affairs, Provention Bio
-
Dr. Francine Kaufman, Distinguished Professor Emerita of Pediatrics, Keck School of Medicine of the University of Southern California; Advisory Board, diaTribe
-
Dr. Felicia Pagliuca, Scientist & Vice President/Disease Area Executive, Type 1 Diabetes, Vertex
-
Dr. Georgea Pasedis, Doctor of Pharmacy & Senior Vice President, Global Head of Medical Affairs, Dompé
Read below for insights from the event.
On the potential of clinical trials
Clinical trials are an important aspect of scientific breakthroughs and discovery. For the latest drugs and technology to make their way to the general public, they first must be rigorously tested to make sure that they are safe and effective. In diabetes, clinical trials play a crucial role – and we wouldn’t have any of the current advancements like AID systems without them. Dr. Pagliuca (Vertex) put this eloquently: “Clinical trials are really the bridge between breakthrough science and development of new medicines.”
Well-run clinical trials take time, but they have the potential to teach us so much about the course of diabetes. As Dr. Kaufman said, “We have to look not just at delaying [the onset of diabetes], but what is the course of the disease after that? Is there less glycemic variability, more Time in Range? These aren’t just two- or three-year studies, these are studies that need to be ongoing over a longer time period.”
As we begin to think about the potential to delay or prevent type 1 diabetes, or even cure it, we know that these treatments will need to be tested in controlled settings. To do that, we need more people who are eligible to enroll in clinical trials and to encourage others to do so as well. “One of my take-home messages is that clinical trials are really a good thing. It’s how we advance science into the clinic, and you should pocket it up, think about it, and encourage others who you know to participate,” said Dr. Moses.
You can learn more about clinical trials for new diabetes treatments, technology, and care programs that may be recruiting people with diabetes in your area here.
On recruitment for clinical trials
Recruiting diverse populations for clinical trials and making sure that all people are aware of the trials they may be eligible for is challenging. The panelists agreed that researchers need to do a better job at recruiting into trials in general, but specifically those from underrepresented and underserved populations into trials.
Dr. Pasedis said one important way of doing this is through “patient-doctor relationships” and technology platforms that can “help us predict enrollment and recruitment patterns.” Others agreed that family care doctors, rather than endocrinologists, are a good place to start, as they are the ones that people seeking care likely trust the most.
The experts shared additional thoughts on the difficult dilemma of recruiting diverse populations.
“We need to make sure that endocrinologists and other healthcare providers are aware of the clinical trials their patients may be candidates for,” Dr. Pagliuca said. “And we need to make sure those who aren't patients at large academic medical centers have the same opportunities to participate in clinical trials. Unfortunately, this is a disease [type 1 diabetes] of more than 1.5 million people in the US alone so there are many people who could benefit from these clinical trials.” It’s important to know that sometimes clinical trials will allow you to travel to their sites (and cover the costs) if you do not have a trial site near you. And many clinical trials have begun using telehealth to expand their reach.
Dr. Pasedis added: “We need to find better ways to recruit patients and expand our recruitment beyond the isolated populations we include. I think about how to target underserved areas. Many clinical trials don’t have the right level of diversity. Let’s talk about the groups that are in the forefront of being included in clinical trials, and how to continue identifying the right patients to be in clinical trials.”
Dr. Anhalt emphasized the value of taking a population-based approach to recruitment. “The people living with the disease who would be most appropriate or benefit the most from clinical trials live in their home, and the clinical trials are happening in the clinic,” he said. “We have to recognize that unless screening is done in a systematic, population-wide fashion to identify individuals who have autoantibodies, we are going to be stuck in this situation over and over again.”
As we learn more about the social determinants of health and health inequity, we need to assure that study populations are representative of all people with diabetes is increasingly crucial. To learn more, read our article: “How Race and Ethnicity Affect Diabetes Prevalence, Management, and Complications.”
On screening for type 1
The panelists were very clear: without screening, we cannot hope to successfully prevent or delay type 1 diabetes. And we shouldn’t limit screening to only those with family members who have the disease. Dr. Moses noted that over 90% of individuals with type 1 have no family history of diabetes, making it harder to properly diagnose, which is where screening can help identify risk. Dr. Anhalt held a similar view. “Those in stage 1 [of type 1 diabetes] have a nearly 100% lifetime risk of developing stage 3,” he said. “Identification of those with stage 1 or stage 2 is dependent on screening, and this highlights one of the most critical reasons for drilling down on those antibodies [that indicate your risk for developing type 1 diabetes].”
Highlighting another important reason for screening, Dr. Anhalt spoke to data from at-risk and general population screening which have demonstrated a significant decrease in the incidence of diabetic ketoacidosis (DKA), a potentially fatal condition, as well as long term negative impact on glycemic outcomes.
However, screening is no small or easy task. “Screening is hard,” Dr. Kaufman said. “People don't necessarily want to know that they are at risk for something like type 1 diabetes.”
Dr. Kaufman elaborated on these challenges: “Of course, going to the general public and screening for something that's still has a very low incident rate isn’t easy. When do we screen, who do we screen, who pays for the screening? It is going to have to be a public health endeavor.”
On preventions and cures
Though we aren’t there yet, the panelists agreed that the possibility of being able to prevent or even cure type 1 diabetes is on the horizon. What it will take to get there is another story. With prevention and delay treatments in the works, there is a need to define what a “cure” really means when it comes to diabetes and, specifically, what it means for people with type 1. Dr. Anhalt explained, “What a cure means for someone is an individual assessment for what treatment someone receives. A cure means different things to different people. One thing I do know is that insulin is not a cure. No one would agree that insulin is a cure.”
Likewise, Dr. Kaufman echoed the challenges of characterizing the meaning of potential cures. “We have to get closer to more and more people getting normalized glucose,” she said. “Would it be a cure if you got to ‘normal’ glucose levels without having to take daily insulin but still have to take immunosuppressants? Someone has to decide that. [I believe at a] minimum, we have to at least get to something that normalizes glucose and other metabolic parameters associated with insulin deficiency and insulin administration that isn’t physiologic.”
Additionally, there is a need to make sure that access to clinical trials and these advancements in treatment reach everyone, especially those that diabetes most adversely affects. “To ensure diabetes treatments are accessible to everyone, we need a much more demanding, sustained commitment, and level of cooperation across the global ecosystem,” Dr. Pasedis said. “We need to holistically bring clinical trials to underrepresented populations who maybe don’t have access to education.”
Dr. Pagliuca said the more we learn and discover, the more hopeful the future seems to become. “What’s really exciting for us is that science really has turned a corner in the last few years in our understanding of beta cell, stem cell biology, immunology and we’re putting those pieces together,” she said. “I think we are closer than we’ve ever been in this type of technology. There is more work to be done of course, but when I look back at what we were able to do ten years ago versus today, it’s really night and day.”
Watch the full event here!