Ozempic Slows Kidney Disease, Cuts Cardiorenal Death Risk in Type 2 Diabetes, FLOW Study Finds
By Susannah Chen
 Semaglutide offered kidney and heart benefits to people with type 2 diabetes and chronic kidney disease, the FLOW trial showed, opening the door for more uses for Ozempic.
Semaglutide offered kidney and heart benefits to people with type 2 diabetes and chronic kidney disease, the FLOW trial showed, opening the door for more uses for Ozempic.
People with type 2 diabetes and chronic kidney disease who took Ozempic (semaglutide) slowed the progression of their kidney disease and cut their risk of death from kidney disease and major cardiac events by 24%, according to new data from Novo Nordisk’s FLOW trial.
Anticipation of the FLOW trial’s results has run high ever since the Danish drugmaker announced that the trial had been halted early based on an interim analysis that treatment was successful.
FLOW kidney outcomes trial: Study design
The FLOW trial was a phase 3 trial of more than 3,500 participants with type 2 diabetes and moderate to severe chronic kidney disease (CKD) in 28 countries. The study, which began in 2019, compared a 1 mg dose of Ozempic (injectable semaglutide) against a placebo as an add-on to the standard treatment for CKD.
The objective of the FLOW trial was to see if Ozempic delayed CKD progression and lowered risk of kidney- and cardiovascular-related death.
When and why did Novo Nordisk end the FLOW trial?
The FLOW trial was cut short in October of last year, one year ahead of schedule, after an interim analysis determined that the initial results were sufficient enough to prove the treatment was effective. Novo Nordisk, the Danish drugmaker behind Ozempic, said it plans to file for a label expansion of Ozempic in the U.S. and in the European Union in 2024.
What were the key findings? Did the FLOW trial meet expectations?
The FLOW trial findings were generally positive, as Ozempic slowed the progression of kidney disease and reduced the risk of death from kidney disease and major cardiac events by 24%. Many industry experts, however, were anticipating that results would demonstrate cardiorenal risk reduction of more than 30%, meaning that the trial fell short of some expectations.
In the trial, Ozempic was generally safe and well tolerated with side effects in line with other trials of the drug.

The trial’s five-part primary endpoint included the following guidelines:
-
A reduction of eGFR of 50% or greater compared with baseline
-
Onset of a persistent eGFR of less than 15
-
The initiation of dialysis or kidney transplantation
-
Death from kidney disease
-
Death from cardiovascular disease
The FLOW trial's secondary outcomes included:
-
Annual rate of change of eGFR
-
Major adverse cardiovascular events (MACE) including non-fatal myocardial infarction, non-fatal stroke, and cardiovascular death
-
All-cause death
What happens now that the FLOW trial is over?
While topline results from the FLOW study have been made public, Novo Nordisk said it expects to present full results at a scientific conference sometime in the first half of 2024.
Given the success of the FLOW trial, Novo Nordisk has said that it expects to file for a label expansion for Ozempic to include the prevention of kidney disease progression in patients with type 2 diabetes and CKD in the U.S. and European Union sometime this year.
What implications does the FLOW trial have for Ozempic and other drugs in its class?
There are a few implications that the results of this trial could have for Ozempic as well as other GLP-1 receptor agonists and incretin drugs in general.
Ozempic and other GLP-1s will likely also become treatment for kidney disease
These positive study results will likely set the stage for Ozempic – and other drugs in the same class, from oral semaglutide to injectable tirzepatide – to become a treatment option for kidney disease in addition to type 2 diabetes and obesity.
“The positive results from FLOW demonstrate the potential for semaglutide to become the first GLP-1 treatment option for people living with type 2 diabetes and chronic kidney disease,” Martin Holst Lange, Novo’s head of development, said.
In addition to Novo Nordisk’s planned label expansion for Ozempic, Eli Lilly is also enrolling patients in its own trial, TREASURE-CKD, to see if tirzepatide (marketed under the names Mounjaro and Zepbound) can treat CKD in people with obesity, both with and without type 2 diabetes.
Successful treatment of CKD with these medications could have implications that are far ranging. Lange estimated that 40% of people who live with type 2 diabetes also live with CKD. Given that an estimated 462 million individuals worldwide are affected by type 2, this means that nearly 185 million people could potentially stand to benefit from medications that improve kidney outcomes.
Ozempic and other GLP-1s will likely be compared to another diabetes drug class, SGLT-2 inhibitors
Given Ozempic’s reduction of kidney disease events in the FLOW study, it seems likely that the GLP-1’s kidney protective benefits will be compared against those of SGLT-2 inhibitor drugs.
SGLT-2 inhibitors, which include Jardiance (empagliflozin) and Farxiga (dapagliflozin), are a different class of diabetes medications that have demonstrated kidney risk reduction. SGLT-2 inhibitors are widely used to guard against progression of kidney disease in adults with CKD, both with and without type 2 diabetes.
From a kidney disease reduction standpoint, SGLT-2s will be tough competitors for Ozempic and other GLP-1s to beat. In comparable trials, SGLT-2 medications all demonstrated a greater reduction in kidney event risk than Ozempic did in the FLOW trial.
In one landmark trial, EMPA-KIDNEY, participants who took Jardiance had a 28% lower risk of progression of kidney disease or death from cardiovascular causes compared to those who didn’t. Another trial, DAPA-CKD, demonstrated a 39% risk reduction with Farxiga. And in the CREDENCE trial, patients with type 2 diabetes and CKD who took canagliflozin (Invokana) reduced their risk of kidney failure and cardiovascular events by 30%.
Could Ozempic and other diabetes drugs reduce the need for dialysis? Not so fast, providers say
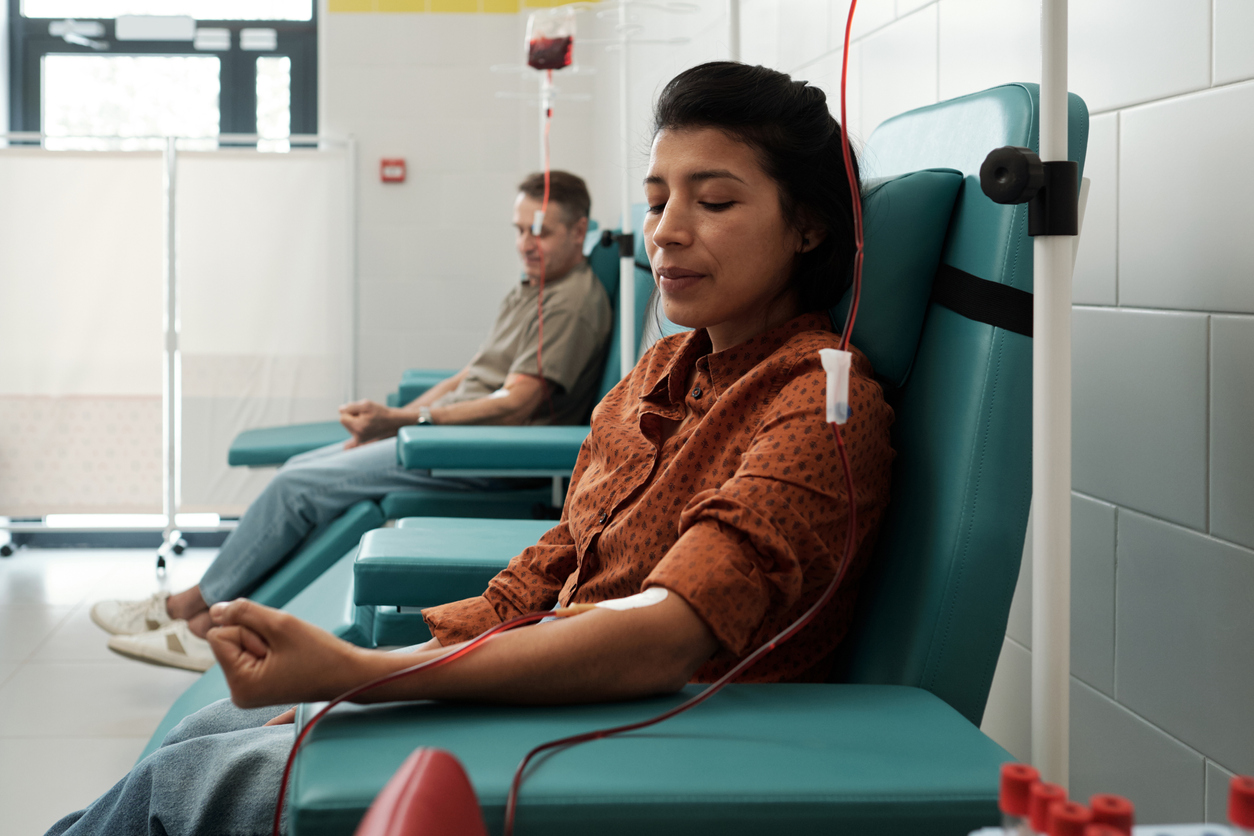 Currently the standard treatment for kidney failure has been dialysis, a process that removes extra fluid and waste from the blood. It’s possible, however, that FLOW and other trials of diabetes drugs on kidney outcomes could potentially reduce the need for dialysis in some patients with CKD. In addition to the FLOW trial, other studies have shown that the SGLT-2 inhibitor drug class slows the progression of kidney disease.
Currently the standard treatment for kidney failure has been dialysis, a process that removes extra fluid and waste from the blood. It’s possible, however, that FLOW and other trials of diabetes drugs on kidney outcomes could potentially reduce the need for dialysis in some patients with CKD. In addition to the FLOW trial, other studies have shown that the SGLT-2 inhibitor drug class slows the progression of kidney disease.
DaVita, one of the largest dialysis providers in the country, has been following such research closely. For now, it has cast doubt on just how far-reaching Ozempic’s therapeutic benefits may be.
“Although it is nearly impossible to draw any conclusions from the FLOW study at this point because the study results have yet to be released, based on the inclusion criteria for study participants, we believe there may be limited application of the FLOW study findings to the overall CKD patient population,” Dr. Jeff Giullian, chief medical officer for DaVita, said.
 He argued that the inclusion requirements necessary to partake in the FLOW trial, such as needing to be on a standard kidney disease treatment and have a certain amount of protein in the urine, were attributable to a narrow subset – less than 10% – of all current CKD patients.
He argued that the inclusion requirements necessary to partake in the FLOW trial, such as needing to be on a standard kidney disease treatment and have a certain amount of protein in the urine, were attributable to a narrow subset – less than 10% – of all current CKD patients.
“Future trials and research will be required to determine whether any findings derived from the FLOW study could benefit a CKD population beyond those studied in the trial,” Giullian said.
Ozempic and other GLP-1s may also become approved to treat conditions beyond kidney disease
Currently, semaglutide is only approved for two conditions: as a once-weekly injection in 0.5 mg, 1 mg, and 2 mg doses as an adjunct to diet and exercise for type 2 diabetes under the Ozempic label, and as a once-weekly injection in 1.7 mg and 2.4 mg doses for obesity under the Wegovy label.
The FLOW trial results are the latest indication, however, that incretin drugs have benefits for conditions beyond their original intended treatments of type 2 diabetes and weight loss. A number of Novo Nordisk-sponsored studies have offered further evidence that medications such as semaglutide can offer additional benefits, including for heart health.
In addition to initial FLOW trial findings indicating Ozempic can offer kidney protective benefits, results of Novo Nordisk’s SELECT trial showed that semaglutide significantly reduced rates of kidney and cardiovascular events compared with placebo in participants with overweight and obesity with cardiovascular disease but without diabetes. In SELECT, Wegovy reduced the risk of MACE by 20% compared to the placebo group.
Novo Nordisk has also been studying semaglutide in metabolic dysfunction-associated steatohepatitis, a type of liver disease also known as nonalcoholic steatohepatitis, or NASH.
Ozempic, already approved for cardiovascular benefits in type 2 patients, may also be be approved for the same use in people with obesity
 Ozempic (semaglutide 0.5 mg, 1 mg, and 2 mg) previously received FDA approval for cardiovascular health in people with type 2 diabetes after combined results from the SUSTAIN-6 and PIONEER-6 trials showed a 24% reduction in major adverse cardiac events (MACE) such as heart attack, stroke, and other cardiovascular-related deaths.
Ozempic (semaglutide 0.5 mg, 1 mg, and 2 mg) previously received FDA approval for cardiovascular health in people with type 2 diabetes after combined results from the SUSTAIN-6 and PIONEER-6 trials showed a 24% reduction in major adverse cardiac events (MACE) such as heart attack, stroke, and other cardiovascular-related deaths.
Following the success of the SELECT trial, Wegovy (semaglutide in doses of 1.7 mg and 2.4 mg) may lead to a similar approval for cardiovascular protection in people with overweight or obesity.
Further reading:
-
Studies Suggest Diabetes Drugs Help Hearts and Kidneys for People With or Without Diabetes
-
Keeping Your Heart and Kidneys Healthy When You Have Diabetes
Photo credits: Susannah Chen (Ozempic); iStock (all other photos).